I have finalised the demo for the ICH-GCP E6 R3 refresher course. Overall, I liked the content and the interface. I also want to thank Whitehall Train...
About
Written by an expert with over 30 years of experience up to Research Director level in a major pharmaceutical group, this GCP training course covers the ICH-GCP (E6 R3) guidelines as adapted for the UK and meets the training requirements for participation in UK and international clinical trials.
Course Syllabus
- The History of GCP: Part 1
- The History of GCP: Part 2
- The History of GCP: Part 3
- The History of GCP: Part 4
- What is GCP?
- How was GCP introduced into the UK?
- The Principles of ICH GCP: Part 1
- The Principles of ICH GCP: Part 2
- Some General Points
- Documentation/Records and Version Control
- Quality Assurance (QA)
- MHRA GCP inspections
- Other Resources
- Introduction: Part 1
- Responsibilities of the CA
- Responsibility of IEC I
- Responsibility of IEC II
- Subject Informed Consent Forms (ICF) I
- Subject Informed Consent Forms (ICF) II
- Composition, Functions, Operations, Procedures and Records
- IEC interactions with Sponsors and Investigators
- Introduction
- Investigator Responsibilities
- Investigator's Qualifications and Agreements
- Adequate Resources
- Medical Care of Trial Subjects I
- Medical Care of Trial Subjects II
- Communication with IRB/IEC I
- Communication with IRB/IEC II
- Compliance with the Protocol
- Investigational Medicinal Product
- Randomization Procedures and Un-blinding
- Informed Consent I
- Informed Consent II - The Consent Discussion
- Informed Consent III - Vulnerable Participants
- Informed Consent IV - Participants who Cannot Read or Write
- Informed Consent IV - Minors and participants who lack capacity
- Informed Consent V - Minors and participants who lack capacity
- Informed Consent VI - Incapacitated Participants
- Informed Consent VII - Incapacitated Participants
- Informed Consent VIII - Updating Consent
- Records and Reports I
- Records and Reports II
- Records and Reports III
- Records and Reports- IV Computerised Systems
- Records and Reports V - Study Site Files
- Records and Reports VI - Updates & Amendments
- Records and Reports VII - Source Documents
- Records and Reports VIII - Source Documents
- Records and Reports IX- Source Documents
- Records and Reports X - Financial Information & Contracts
- Records and Reports XI - Financial Information & Contracts
- Records and Reports XII - The Case Record Form (CRF)
- Records and Reports XIII - Recording Subject Data
- Premature Termination or Suspension of a Trial
- Progress & Final Reports by Investigator
- Archiving/Retention of Essential Records
- Introduction: Part 1
- Introduction: Part 2
- Quality Management I
- Quality Management II
- Quality Assurance and Quality Control I
- Quality Assurance and Quality Control II - SOPs
- Quality Assurance and Quality Control III - Agreements & Contracts
- Quality Assurance and Quality Control IV - Agreements & Contracts
- Service Providers
- Contract Research Organisations
- Trial Design
- Trial Management I
- Trial Management II - Data Management
- Trial Management III - Electronic Data Systems
- Trial Management IV - Record Keeping
- Investigator Selection I
- Investigator Selection II - Permissions
- Investigator Selection III - Responsibilities
- Investigator Selection IV - Compensation
- Financing I
- Financing II
- Notification/Submission to Regulatory Authorities
- Gaining Regulatory Authority approval in the UK
- Confirmation of Review by IRB/IEC
- Manufacturing, Packaging, Labelling and Coding Investigational Products: Part 1
- Manufacturing, Packaging, Labelling, and Coding Investigational Products: Part 2
- Supplying and Handling Investigational Products
- Record Access
- Data Handling I
- Data Handling II
- Data Handling III
- Data Handling IV
- Data Handling V
- Data Handling VI
- Statistical Programming and Data Analysis I
- Statistical Programming and Data Analysis II
- Record Keeping and Retention
- Audit and Inspection I
- Audit and Inspection II
- Noncompliance
- Premature Termination or Suspension of a Trial: Part 1
- Premature Termination or Suspension of a Trial: Part 2
- Clinical Trial/Study Reports
- Multicentre Trials
- Introduction
- Data Governance Part 1
- Data Governance Part 2
- Maintain the Blind
- Data Life Cycle I
- Data Life Cycle II
- Data Life Cycle III
- Data Life Cycle IV
- Computerised Systems I
- Computerised Systems II
- Computerised Systems III
- Computerised Systems IV
- Computerised Systems V
- Computerised Systems VI
- Introduction
- Monitor
- The Monitoring Visit: Part 1
- The Monitoring Visit: Part 2
- Verifying IMP
- Compliance
- Verifying Consent
- The Case Record Form (CRF) & Source Documents
- Verifying Participant Data I
- Verifying Participant Data II
- Closing out the Monitoring Visit
- The Monitoring Report & Plan
- Quality Management - Centralised Monitoring
- Fraud and Misconduct: Part 1
- Fraud and Misconduct: Part 2
- Introduction
- AEs, ADRs, & SUSARs
- SAEs and Serious ADRs
- SUSARs
- Adverse Events of Special Interest
- Periodic Safety Reports
- Reporting Decision Tree
- Introduction
- Protocol Structure and Content: Part 1
- Protocol Structure and Content: Part 2
- Treatment of Participants
- Introduction
- Table of Contents of Investigator's Brochure (Example)
- Essential Records
- Archiving
- Essential Records
- Documents to be Present Pre-Study
- Documents to be Added During the Study
- MHRA UK-Specific ICH E6(R3)
- Regulatory Context: The Hierarchy of Compliance
- Applicability Statement: Scope & Binding Status
- Proportionality & Risk-Based Quality Management Theme: Designing quality In (Quality by Design)
- Sponsor Oversight & Delegation Theme: Clear Roles & Responsibilities
- Vendor Management & Contracts Theme: Supply chain integrity & Service providers
- Data Governance, Integrity & UK GDPR Theme: Protecting Data & Participant Rights
- Essential Records & TMF Expectations Theme: Reconstructing the Trial
- Technology, Digital Systems & Validation Theme: Computerized Systems Validation (CSV)
- Safety Reporting & Pharmacovigilance Alignment Theme: Reporting Lines (The “UK Divergence”)
- Inspection Focus: The ‘Story’ of the Trial
- Common Misinterpretations: ICH Text vs. UK Law
- Evidence & Demonstration Theme: Compliance is Demonstrated, Not Stated
- Key Takeaways for UK Compliance
- Glossary & Abbreviations
- EU Guidance Documents
- ICH Guidance Documents
- US FDA Guidance Documents
- GCP Course Printouts
- Global Competent Authorities List
Our Certified Customers
Learner Rating & Reviews
Frequently Asked Questions
Good Clinical Practice (GCP) training is an essential educational program that equips researchers and clinical trial professionals with the knowledge of ethical and scientific standards crucial for conducting high-quality clinical trials. This comprehensive training covers the internationally recognized guidelines established by the International Council for Harmonisation (ICH).
The primary goals of GCP training are:
- Ensuring the protection of human subjects' rights, safety, and well-being
- Maintaining the integrity and reliability of clinical trial data
- Promoting consistent, high-quality practices across all aspects of clinical research
Our GCP course delves into these critical areas, providing learners with a solid foundation in the principles and practical applications of Good Clinical Practice.
For researchers interested in participating in clinical trials, GCP certification is a necessity.
For those who are simply interested in improving their understanding of the field, GCP certification is highly valuable due to its:
- Ensures compliance with international standards
- Enhances research credibility and quality
- Protects participant rights and safety
- Improves career prospects in clinical research
Our course not only provides certification but also equips you with practical skills to apply GCP principles effectively in your work.
GCP certification is essential for a wide range of professionals in clinical research:
- Clinical Trial Investigators: Principal investigators and sub-investigators responsible for trial conduct at research sites.
- Clinical Trial Staff: Including research coordinators, study nurses, and other site personnel involved in trial management.
- Sponsors and Contract Research Organizations (CROs): Those overseeing trial planning, initiation, and reporting.
- Regulatory Authorities: Officials who monitor and evaluate trial compliance.
- Institutional Review Boards (IRBs) and Ethics Committees: Members reviewing and approving trial protocols.
- Academic and Research Institution Staff: Ensuring adherence to international standards in institutional research.
- NIH-Funded Researchers: All investigators and staff involved in NIH-funded clinical trials.
Our course caters to this diverse audience, providing role-specific insights alongside core GCP principles. The course is also valuable for anyone looking to upskill their research abilities and improve their clinical trials operations.
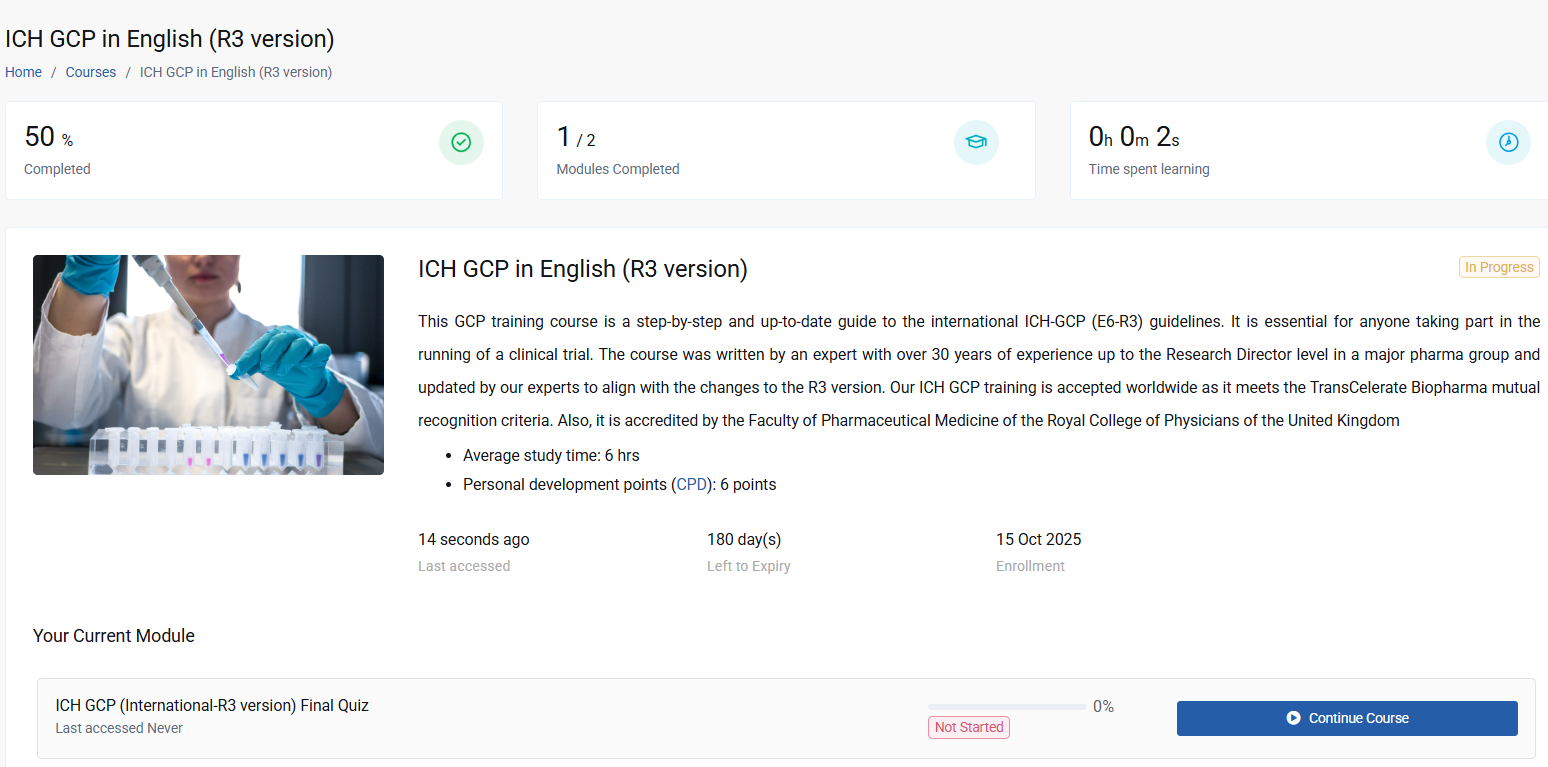
This GCP training course covers the ICH-GCP (E6-R3) international guidelines and meets the training requirement for you to participate in international clinical trials. Multiple language versions are available. This GCP training course is a step-by-step and up-to-date guide to the international ICH-GCP (E6-R3) guidelines. It is essential for anyone taking part in the running of a clinical trial.
The course was written by an expert with over 30 years of experience up to the Research Director level in a major pharma group. Our ICH GCP training is accepted worldwide as it meets the TransCelerate Biopharma mutual recognition criteria. Also, it is accredited by the Faculty of Pharmaceutical Medicine of the Royal College of Physicians of the United Kingdom. Apart from English, the course is available in German, Bulgarian, French, Italian, Japanese, Polish, Portuguese, Russian and Spanish. We also produce regional versions of this course tailored to the specific regulatory frameworks in Australia, the UK, the US, France, Germany and Latin America.
This course features a clear and visually appealing format, allowing for easy cross-referencing to the ICH-GCP E6 document. It offers valuable insights into the practical application of Good Clinical Practice (GCP) based on the author's extensive experience. Furthermore, it is accredited by the Faculty of Pharmaceutical Medicine of the Royal College of Physicians of the United Kingdom and provides participants with the opportunity to earn 6 CPD points.
Yes, our GCP course is accredited by two industry-leading organisations:
- TransCelerate Biopharma Inc.: A nonprofit organisation collaborating with 20 major pharmaceutical companies. Their mutual recognition program is considered the gold standard in the field of clinical practice.
- The Faculty of Pharmaceutical Medicine at the Royal College of Physicians: The professional membership body for pharmaceutical physicians in the UK, known for setting rigorous standards for research since 1989.
These accreditations ensure our course meets the highest industry and academic standards, offering you a widely recognized certification.
Costs vary depending on the following factors:
- Accreditation: Is the course approved by official organisations, like TransCelerate?)
- Certification: Does the course meet the ICH requirements that allows researchers to participate in international clinical trials?
- Quality of content: Is the course up-to-date, and written by an expert?
- Access: How long are learners able to access the course?
Whitehall Training’s GCP course is priced due to its:
- Accreditation: It is TransCelerate approved, and accredited by the Royal College of Physicians with 6 CPD points.
- Certification: The course enables users to participate in clinical trials, following the ICH E6(R3) guidelines.
- Quality of content: Our course is written by our Good Clinical Practice expert, Lucy Parker, who has over a decade of experience directing research across large research institutes such as the NHS.
- Access: To support the lifelong learning of our researchers, we provide lifetime access to our course resources.
Buying for a team? We offer 10% off orders of 5 licences at checkout. For discounts on larger orders, please get in touch with our team.