Jeg har færdiggjort demoen for ICH-GCP E6 R3 opfriskningskurset. Overordnet set kunne jeg godt lide indholdet og brugerfladen. Jeg vil også gerne takk...
Om
Dette GCP-træningskursus er skrevet af en ekspert med over 30 års erfaring op til forskningsdirektørniveau i en større farmaceutisk gruppe. Det dækker de internationale ICH-GCP (E6-R3) retningslinjer og opfylder træningskravene for at deltage i internationale kliniske forsøg.
Kursusplan
- GCP's historie: Del 1
- GCP's historie: Del 2
- GCP's historie: Del 3
- GCP's historie: Del 4
- Hvad er GCP?
- Principperne for ICH GCP: Del 1
- Principperne for ICH GCP: Del 2
- Yderligere læringspunkter
- Dokumentation og versionskontrol
- Kvalitetssikring (QA)
- Nøgleressourcer: Del 1
- Nøgleressourcer: Del 2
- Introduktion: Del 1
- Introduktion: Del 2
- Introduktion: Del 3
- Introduktion: Del 4
- Introduktion: Del 5
- Introduktion: Del 6
- Introduktion: Del 7
- Introduktion: Del 8
- Introduktion: Del 9
- Tilsynsmyndighedens ansvar
- IEC's ansvar
- Informerede samtykkeerklæringer for forsøgspersoner (ICF) Del 1
- Informerede samtykkeerklæringer for forsøgspersoner (ICF): Del 2
- Sammensætning, funktioner, operationer, procedurer og optegnelser
- IEC's interaktioner med sponsorer og efterforskere
- Indledning
- Efterforskerens ansvar
- Efterforskerens kvalifikationer og aftaler
- Tilstrækkelige ressourcer
- Lægebehandling af forsøgspersoner: Del 1
- Lægebehandling af forsøgspersoner: Del 2
- Kommunikation med IRB/IEC
- Overholdelse af protokollen
- Forsøgsmedicinske produkter
- Randomiseringsprocedurer og afblinding
- Informeret samtykke: Introduktion
- Informeret samtykke: Samtykkediskussionen
- Informeret samtykke: Personer, der ikke kan læse eller skrive
- Informeret samtykke: Mindreårige og 'mentalt inkompetente' personer
- Informeret samtykke: Ude af stand til at blive behandlet
- Informeret samtykke: Opdatering af samtykke
- Optegnelser og rapporter: Introduktion
- Optegnelser og rapporter: Studiestedsfiler
- Optegnelser og rapporter: Opdateringer og ændringer
- Optegnelser og rapporter: Kildedokumenter
- Optegnelser og rapporter: Finansielle oplysninger
- Optegnelser og rapporter: Sagsjournalformularen
- Optegnelser og rapporter: Registrering af forsøgspersondata
- For tidlig afslutning eller suspension af et forsøg
- Statusrapporter og endelige rapporter fra efterforsker
- Efterforskerens ansvar
- Introduktion: Del 1
- Introduktion: Del 2
- Introduktion: Del 3
- Kvalitetsstyring: Del 1
- Kvalitetsstyring: Del 2
- Kvalitetsstyring Del 3
- QA og QC (kvalitetssikring og kvalitetskontrol): Introduktion
- QA og QC (kvalitetssikring og kvalitetskontrol): Standardprocedurer
- QA og QC (kvalitetssikring og kvalitetskontrol): Aftaler og kontrakter
- Kontraktforskningsorganisationer
- Forsøgsdesign
- Forsøgsstyring: Introduktion
- Forsøgsstyring: Datastyring
- Forsøgsstyring: Elektroniske data
- Forsøgsstyring: Journalføring
- Udvælgelse af efterforsker: Introduktion
- Udvælgelse af efterforsker: Tilladelser
- Udvælgelse af efterforsker: Ansvar
- Udvælgelse af efterforsker: Aflønning
- Finansiering
- Meddelelse/indsendelse til regulerende myndigheder
- Bekræftelse af gennemgang foretaget af IRB/IEC
- Information om IMP
- Fremstilling, emballering, mærkning og kodning af testprodukter: Del 1
- Fremstilling, emballering, mærkning og kodning af testprodukter: Del 2
- Levering og håndtering af afprøvningsprodukter
- Optag adgang
- Datahåndtering I
- Datahåndtering II
- Datahåndtering III
- Datahåndtering IV
- Datahåndtering V
- Datahåndtering VI
- Statistisk programmering og dataanalyse I
- Statistisk programmering og dataanalyse II
- Registrering og opbevaring
- Revision og inspektion
- Manglende overholdelse
- For tidlig afslutning eller suspension af et forsøg: Del 1
- For tidlig afslutning eller suspension af et forsøg: Del 2
- Rapporter fra kliniske forsøg/studier
- Multicenterforsøg
- Indledning
- Datastyring Del 1
- Datastyring Del 2
- Vedligehold blinde
- Datalivscyklus I
- Datalivscyklus II
- Datalivscyklus III
- Datalivscyklus IV
- Computersystemer I
- Computersystemer II
- Computersystemer III
- Computersystemer IV
- Computersystemer V
- Computersystemer VI
- Computersystemer VII
- Indledning
- Overvåge
- Overvågningsbesøget: Del 1
- Overvågningsbesøget: Del 2
- Verifikation af IMP
- Overholdelse af protokollen, ændringerne, standardprocedurerne og vejledningen
- Verifikation af informeret samtykke
- Sagsregistreringsformularen (CRF) og kildedokumenter
- Verifikation af forsøgspersondata
- Afslutning af overvågningsbesøg
- Overvågningsrapport og -plan
- Kvalitetsstyring - Centraliseret overvågning
- Svig og misligholdelse: Del 1
- Svig og misligholdelse: Del 2
- Indledning
- Bivirkninger, bivirkninger og uventede alvorlige bivirkninger
- Alvorlige bivirkninger
- Uventede alvorlige bivirkninger
- Bivirkninger af særlig interesse
- Periodisk sikkerhedsrapport
- Introduktion
- Protokolstruktur og indhold: Del 1
- Protokolstruktur og indhold: Del 2
- Protokolstruktur og indhold: Del 3
- Indledning
- Struktur og indhold af efterforskerbrochuren
- Introduktioner
- Arkivering
- Dokumenter, der skal være til stede før undersøgelsen
- Dokumenter, der skal være til stede efter undersøgelsen
- Ordliste og forkortelser
- EU-vejledningsdokumenter
- ICH-vejledningsdokumenter
- Amerikanske FDA-vejledningsdokumenter
- Udskrifter af GCP-kurser
- Liste over globale kompetente myndigheder
Vores certificerede kunder
Kursistbedømmelser og anmeldelser
Ofte stillede spørgsmål
Good Clinical Practice (GCP) training is an essential educational program that equips researchers and clinical trial professionals with the knowledge of ethical and scientific standards crucial for conducting high-quality clinical trials. This comprehensive training covers the internationally recognized guidelines established by the International Council for Harmonisation (ICH).
The primary goals of GCP training are:
- Ensuring the protection of human subjects' rights, safety, and well-being
- Maintaining the integrity and reliability of clinical trial data
- Promoting consistent, high-quality practices across all aspects of clinical research
Our GCP course delves into these critical areas, providing learners with a solid foundation in the principles and practical applications of Good Clinical Practice.
For researchers interested in participating in clinical trials, GCP certification is a necessity.
For those who are simply interested in improving their understanding of the field, GCP certification is highly valuable due to its:
- Ensures compliance with international standards
- Enhances research credibility and quality
- Protects participant rights and safety
- Improves career prospects in clinical research
Our course not only provides certification but also equips you with practical skills to apply GCP principles effectively in your work.
GCP certification is essential for a wide range of professionals in clinical research:
- Clinical Trial Investigators: Principal investigators and sub-investigators responsible for trial conduct at research sites.
- Clinical Trial Staff: Including research coordinators, study nurses, and other site personnel involved in trial management.
- Sponsors and Contract Research Organizations (CROs): Those overseeing trial planning, initiation, and reporting.
- Regulatory Authorities: Officials who monitor and evaluate trial compliance.
- Institutional Review Boards (IRBs) and Ethics Committees: Members reviewing and approving trial protocols.
- Academic and Research Institution Staff: Ensuring adherence to international standards in institutional research.
- NIH-Funded Researchers: All investigators and staff involved in NIH-funded clinical trials.
Our course caters to this diverse audience, providing role-specific insights alongside core GCP principles. The course is also valuable for anyone looking to upskill their research abilities and improve their clinical trials operations.
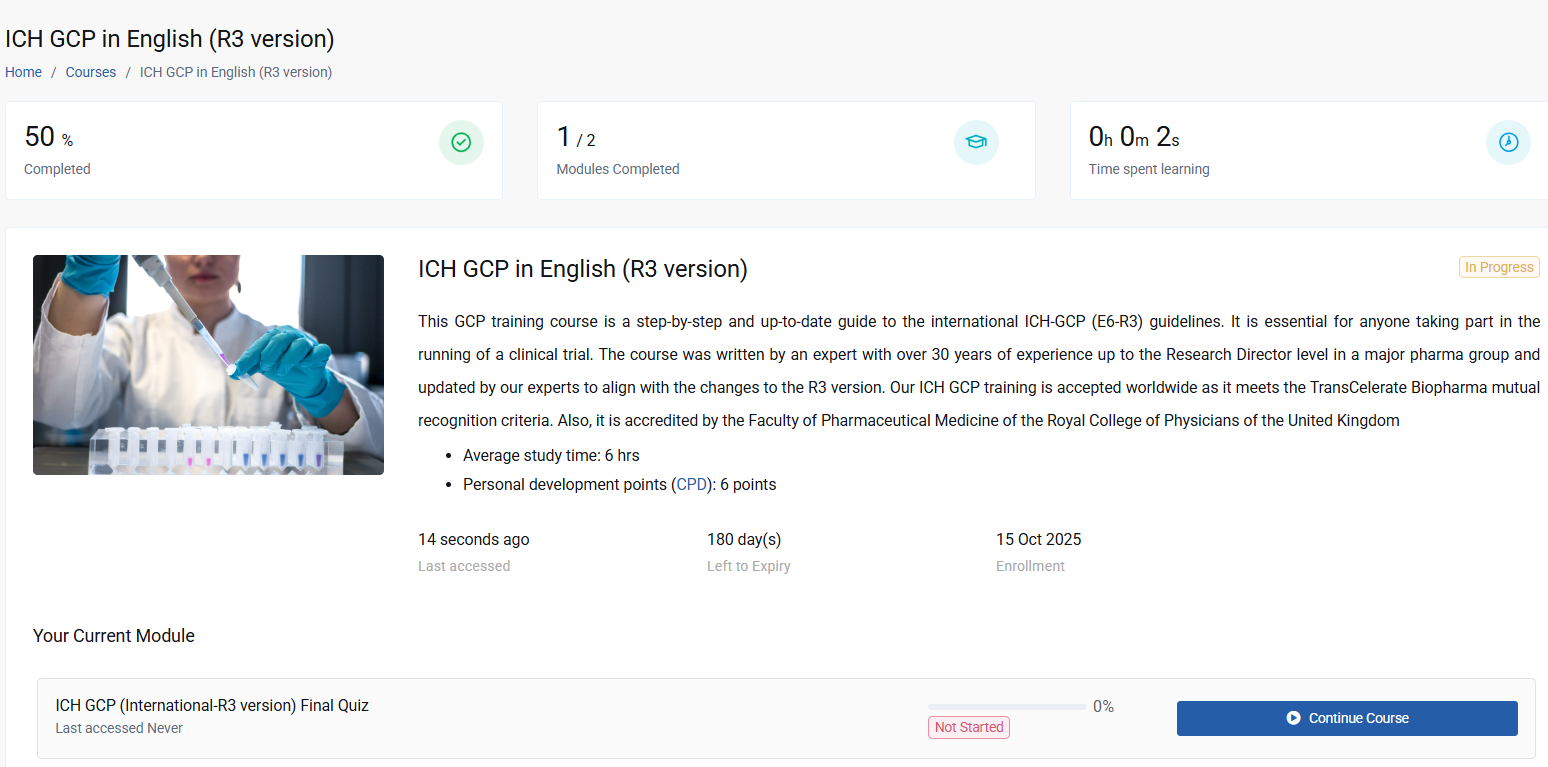
This GCP training course covers the ICH-GCP (E6-R3) international guidelines and meets the training requirement for you to participate in international clinical trials. Multiple language versions are available. This GCP training course is a step-by-step and up-to-date guide to the international ICH-GCP (E6-R3) guidelines. It is essential for anyone taking part in the running of a clinical trial.
The course was written by an expert with over 30 years of experience up to the Research Director level in a major pharma group. Our ICH GCP training is accepted worldwide as it meets the TransCelerate Biopharma mutual recognition criteria. Also, it is accredited by the Faculty of Pharmaceutical Medicine of the Royal College of Physicians of the United Kingdom. Apart from English, the course is available in German, Bulgarian, French, Italian, Japanese, Polish, Portuguese, Russian and Spanish. We also produce regional versions of this course tailored to the specific regulatory frameworks in Australia, the UK, the US, France, Germany and Latin America.
This course features a clear and visually appealing format, allowing for easy cross-referencing to the ICH-GCP E6 document. It offers valuable insights into the practical application of Good Clinical Practice (GCP) based on the author's extensive experience. Furthermore, it is accredited by the Faculty of Pharmaceutical Medicine of the Royal College of Physicians of the United Kingdom and provides participants with the opportunity to earn 6 CPD points.
Yes, our GCP course is accredited by two industry-leading organisations:
- TransCelerate Biopharma Inc.: A nonprofit organisation collaborating with 20 major pharmaceutical companies. Their mutual recognition program is considered the gold standard in the field of clinical practice.
- The Faculty of Pharmaceutical Medicine at the Royal College of Physicians: The professional membership body for pharmaceutical physicians in the UK, known for setting rigorous standards for research since 1989.
These accreditations ensure our course meets the highest industry and academic standards, offering you a widely recognized certification.
Costs vary depending on the following factors:
- Accreditation: Is the course approved by official organisations, like TransCelerate?)
- Certification: Does the course meet the ICH requirements that allows researchers to participate in international clinical trials?
- Quality of content: Is the course up-to-date, and written by an expert?
- Access: How long are learners able to access the course?
Whitehall Training’s GCP course is priced due to its:
- Accreditation: It is TransCelerate approved, and accredited by the Royal College of Physicians with 6 CPD points.
- Certification: The course enables users to participate in clinical trials, following the ICH E6(R3) guidelines.
- Quality of content: Our course is written by our Good Clinical Practice expert, Lucy Parker, who has over a decade of experience directing research across large research institutes such as the NHS.
- Access: To support the lifelong learning of our researchers, we provide lifetime access to our course resources.
Buying for a team? We offer 10% off orders of 5 licences at checkout. For discounts on larger orders, please get in touch with our team.